XPS PEAK LOCATIONS SOFTWARE
11, 18– 20 Although the information needed to make useful and reproducible XPS measurements is available, a significant number of new and less experienced XPS users (and often journal reviewers) are fooled by the easy use of instrumental software and are often not aware of the steps and care needed to produce reliable XPS data for specific analytical purposes. Practical guides for effective and efficient XPS analyses have been developed by ASTM Committee E-42 on Surface Analysis 16, 17 and by Technical Committee 201 (TC 201) of the International Organization for Standardization. Through the efforts of many people including instrument vendors, researchers, national standards laboratories and standards committees, it is now possible to operate instruments in highly reproducible and reliable ways. Interlaboratory-comparison studies conducted starting in the late 1970s 14, 15 demonstrated that XPS measurements, first on materials commonly used as catalysts and then on metals, were not reproducible from laboratory to laboratory. Reproducibility issues related to both instrument design and operation and experimental procedures are not new. There are many steps involved in appropriately setting up and verifying instrument performance for a particular experimental objective, choosing suitable data-acquisition strategies, extracting the desired information from what can be complex data, and preparing a satisfactory report. These issues are sometimes complicated by historic differences in instrument capabilities and calibration, and the spread of binding-energy information in available XPS databases. It is clear to experienced XPS users that in many publications where XPS use is reported, the information is limited in some way and that too often the XPS data reported are incomplete or misinterpreted.

11, 12 To a significant degree, this concern has become reality. It was recognized about 20 years ago that, as XPS matured, the reliable use of XPS would likely be constrained by the availability of XPS experts and expertise among the many users of the technique. 7, 9, 10 The rapid growth in the use of XPS is due to the importance of surfaces, very thin films, and interfaces in many areas of science and technology, the ease of operation of XPS instruments, the perceived simplicity of data interpretation, the ability to analyze a wide variety of samples and the provision of desired information. 5 XPS has become essential for many types of research, expanding from chemistry and materials science into many other areas including those associated with environmental, 6, 7 atmospheric, 8 and biological systems.
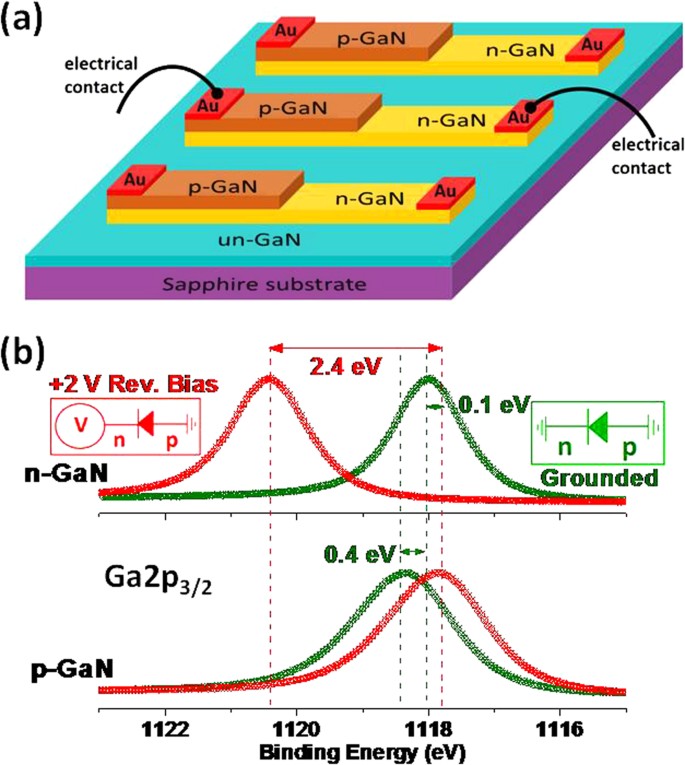
Over the past three decades, the use of X-ray photoelectron spectroscopy (XPS) has grown and it is now the most commonly applied method of surface analysis.
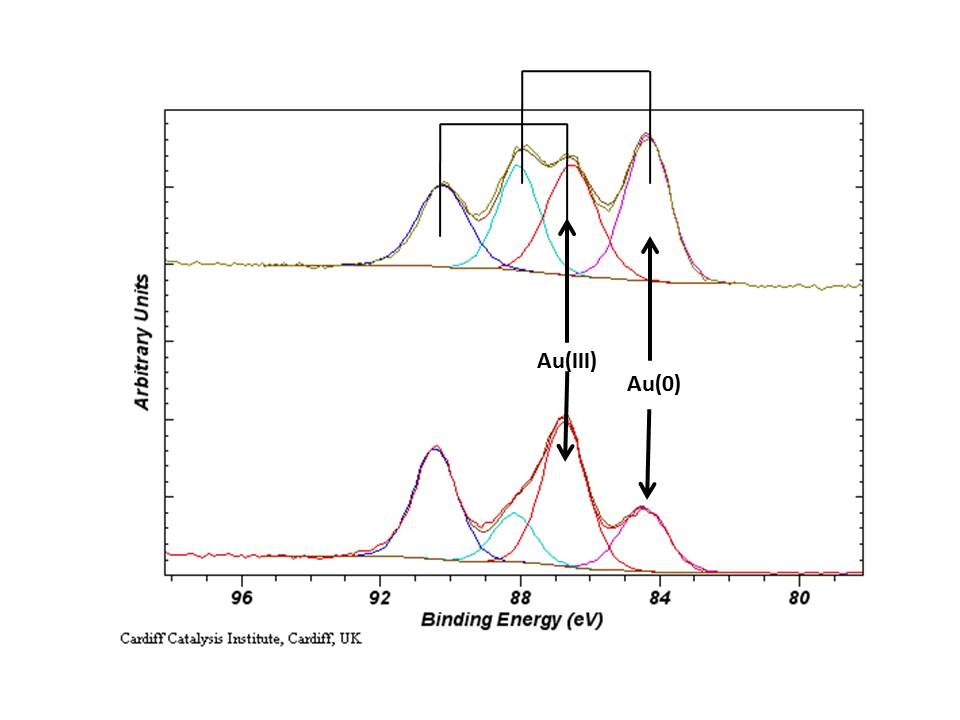
1– 4 The nature and causes of poor reproducibility appear to have many sources, but among them is the availability of a growing suite of automated or semi-automated experimental and computational tools in many research projects. 1 Although such problems are generally thought to exist mainly in clinical or psychological studies, there is evidence that there are significant issues related to reproducibility and replication in most areas of science including those of importance to the American Vacuum Society (AVS).

More than half of the scientists responding to a survey that was published in 2016 indicated that there was a “significant reproducibility crisis” in science and another 38 % indicated that there was a slight reproducibility crisis.
